Molecular geometry table 7.42/26/2024 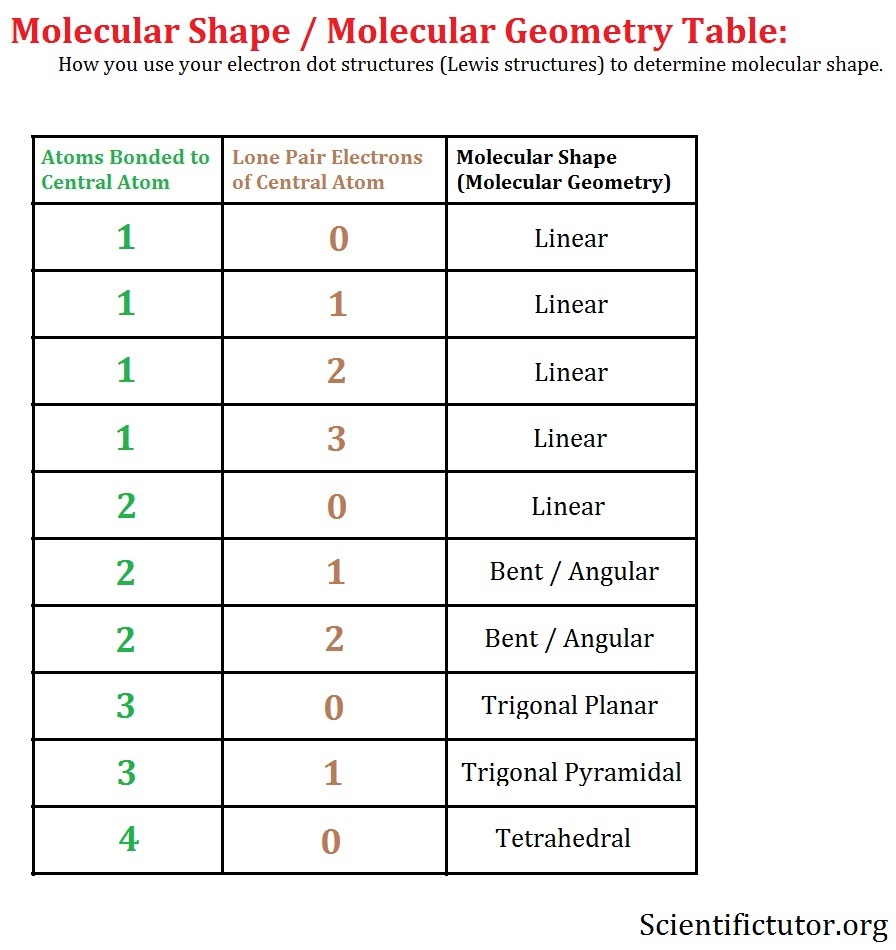
Lone pairs, unpaired electrons, and single, double, or triple bonds are used to indicate where the valence electrons are located around each atom in a Lewis structure. 7.3: Lewis Symbols and Structures Valence electronic structures can be visualized by drawing Lewis symbols (for atoms and monatomic ions) and Lewis structures (for molecules and polyatomic ions).The ability of an atom to attract a pair of electrons in a chemical bond is called its electronegativity. In polar covalent bonds, the electrons are shared unequally, as one atom exerts a stronger force of attraction on the electrons than the other. In pure covalent bonds, the electrons are shared equally. 7.2: Covalent Bonding Covalent bonds form when electrons are shared between atoms and are attracted by the nuclei of both atoms.The charges of anions formed by the nonmetals may also be readily determined because these ions form when nonmetal atoms gain enough electrons to fill their valence shells. The charges of cations formed by the representative metals may be determined readily because, with few exceptions, the electronic structures of these ions have either a noble gas configuration or a completely filled electron shell. 
7.1: Ionic Bonding Atoms gain or lose electrons to form ions with particularly stable electron configurations.They were guided by bonding theory-the topic of this chapter-which explains how individual atoms connect to form more comp But it was not until 1985 that a new form of carbon was recognized: buckminsterfullerene, commonly known as a “buckyball.” Experimental evidence revealed the formula, C60, and then scientists determined how 60 carbon atoms could form one symmetric, stable molecule. 7.0: Prelude to Chemical Bonding and Molecular Geometry It has long been known that pure carbon occurs in different forms (allotropes) including graphite and diamonds.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
